Misunderstanding peptide terminology doesn't just make you sound uninformed at the gym. It can lead you to reconstitute at the wrong concentration, mistime your dosing cycle, or unknowingly purchase a research compound labeled as something else entirely. The peptide space has its own dense lexicon, and key industry terms like lyophilized, reconstitution, half-life, and titration carry real clinical weight. Knowing what these words actually mean, not just recognizing them, is what separates users who see consistent results from those who keep troubleshooting. This guide breaks it all down with practical depth.
Table of Contents
- Core peptide terminology explained
- Common peptide types and classifications
- Usage methodologies: dosing, injection routes, and stacking
- Regulatory and safety terms everyone should know
- Our perspective: where peptide terminology trips up even advanced users
- Get expert guidance for smarter peptide use
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Know your peptide terms | Accurate definitions let you avoid misuse and maximize results from peptide therapy. |
| Class matters | FDA-approved, research-only, and compounded peptides differ greatly in safety and legality. |
| Method shapes outcome | Understanding dosing, stacking, and titration ensures your peptide strategy is both effective and lower risk. |
| Stay evidence-based | New peptides and terms emerge fast; always check regulatory sources before starting a new protocol. |
Core peptide terminology explained
With the stage set for why terms matter, let's decode the key terminology you'll encounter with peptides.
The four foundational terms every user needs to internalize are lyophilized, reconstitution, half-life, and titration. These aren't just vocabulary words. They directly govern how you store your peptides, how you prepare them, how often you inject, and how you ramp up dosing. Getting any one of them wrong can compromise an entire protocol.
Lyophilized refers to a peptide that has been freeze-dried into a stable powder form. This process removes water through sublimation under vacuum, which dramatically extends shelf life and prevents degradation. Most research-grade and compounded peptides you'll encounter come in lyophilized form for exactly this reason. Stability is the goal.

Reconstitution is the process of dissolving that freeze-dried powder in a sterile liquid, almost always bacteriostatic water (water preserved with 0.9% benzyl alcohol to prevent bacterial growth). The ratio you use during reconstitution determines your final concentration, which affects every dose you draw afterward. A common reconstitution error is using too much or too little solvent, throwing off dosing calculations entirely.
Half-life is the time it takes for your body to reduce the active concentration of a peptide by half. This determines dosing frequency. Semaglutide, for instance, carries a half-life of about 7 days, which is why it's dosed weekly. Ipamorelin, by contrast, has a half-life of roughly 2 hours, requiring multiple daily injections to maintain meaningful serum levels. Ignoring half-life means your dosing schedule is essentially random.
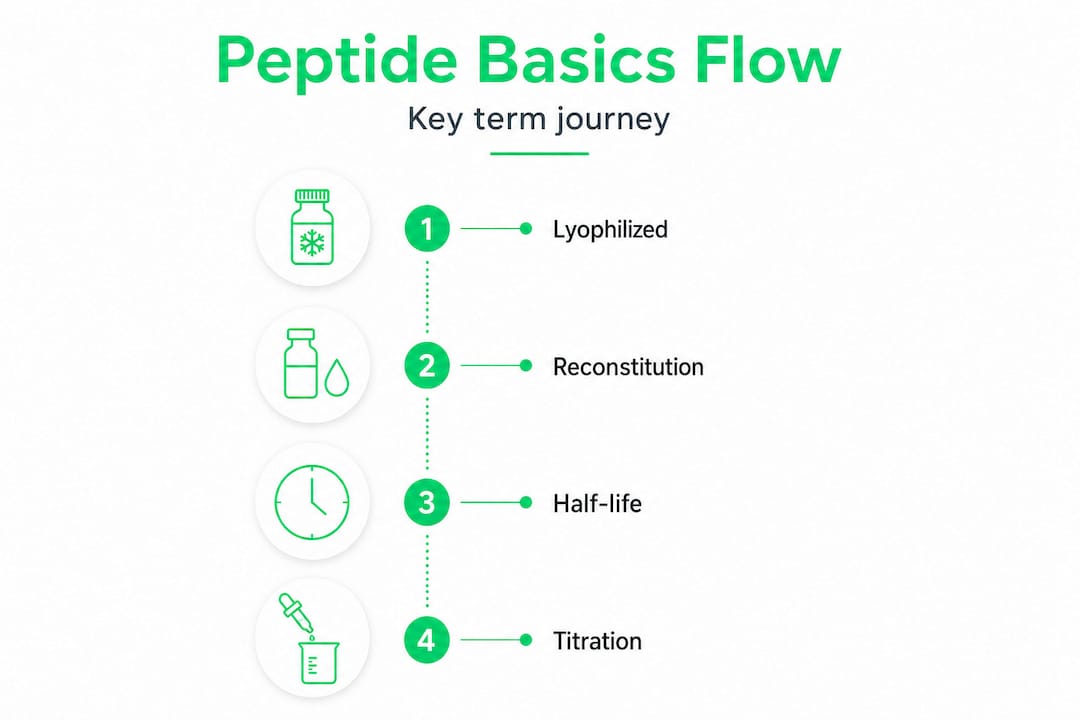
Titration is the practice of starting at a lower dose and gradually increasing toward a therapeutic target. This approach reduces side effects, allows the body to adapt, and helps you identify the lowest effective dose before pushing higher.
| Term | Plain-language meaning | Why it matters in practice |
|---|---|---|
| Lyophilized | Freeze-dried powder form | Ensures stability before use |
| Reconstitution | Mixing powder with bacteriostatic water | Determines final dose concentration |
| Half-life | Time for plasma levels to halve | Drives dosing frequency decisions |
| Titration | Gradual dose escalation | Minimizes side effects and optimizes tolerance |
Two terms that frequently cause confusion are "therapeutic peptide" and "research peptide." These are not interchangeable. A therapeutic peptide has been through clinical trials and received regulatory approval for a specific use. A research peptide has not, and its label will typically state it is not intended for human use. Mixing up these terms is more than a semantic mistake. It has legal and safety implications covered in detail later in this article.
Pro Tip: Always check that your peptide arrives in lyophilized form with intact packaging. Reconstituting a peptide that has already been exposed to moisture or heat before you receive it means you're starting with degraded product, regardless of what the label says.
Learning peptide therapy basics and then connecting them to these foundational terms gives your protocol a measurable edge over trial-and-error approaches.
Common peptide types and classifications
Understanding these foundational terms sets you up to better navigate the various peptide options on the market.
Most peptide users encounter a shortlist of names before they understand what class each peptide belongs to or why that classification affects their stack design. Let's fix that. Detailed peptide classifications matter because they tell you the mechanism, the research depth, and the risk profile simultaneously.
Common peptide names and classes include BPC-157 (a gastric pentadecapeptide studied for tissue repair), TB-500 or Thymosin Beta-4 (a systemic healing peptide), GHK-Cu (a copper tripeptide examined for skin and wound healing), GLP-1 agonists like semaglutide and tirzepatide (used for weight management and metabolic health), and GHS or growth hormone secretagogues like ipamorelin (which stimulate the body's own GH release).
| Peptide | Class | Primary use | Regulatory status |
|---|---|---|---|
| BPC-157 | Gastric pentadecapeptide | Tissue and gut repair | Research only (preclinical) |
| TB-500 | Thymosin peptide | Systemic healing, inflammation | Research only |
| GHK-Cu | Copper tripeptide | Skin healing, anti-aging | Cosmetic and research use |
| Semaglutide | GLP-1 agonist | Weight loss, metabolic health | FDA-approved (Ozempic, Wegovy) |
| Ipamorelin | GHS | Growth hormone stimulation | Research only |
| CJC-1295 | GHRH analog | GH pulse amplification | Research only |
Knowing which bucket a peptide falls into changes everything about how you source it, how you talk to a physician about it, and what outcomes you can realistically expect.
Some of the most popular performance peptide examples exist within the GHS category. Ipamorelin combined with CJC-1295 is a frequently used stack for individuals focused on body composition and recovery, because the two peptides act synergistically. Ipamorelin triggers a clean GH pulse while CJC-1295 extends the amplitude of that pulse. Neither is FDA-approved for these purposes, which shapes the risk profile.
Popular stacks and what their combination means in practice:
- BPC-157 + TB-500 ("Wolverine stack"): Paired for accelerated recovery from musculoskeletal injury; both act through overlapping but distinct healing pathways
- Ipamorelin + CJC-1295: A GHS combination used for lean mass and sleep quality; timing matters significantly here
- GHK-Cu + BPC-157: An emerging combination targeting wound healing and inflammation, though human data remains limited
- Semaglutide + BPC-157: Some users combine a GLP-1 agonist with BPC-157 to counteract GI side effects, though this is highly experimental
Tracking biomarkers while running any of these stacks is increasingly important. Following peptide biomarkers specific to your goals helps you distinguish actual peptide-driven improvement from placebo or confounding lifestyle factors.
Usage methodologies: dosing, injection routes, and stacking
Having a grasp on what the peptides are, let's turn to the practicalities. How they're actually used and why terms about methodology deeply affect safety and results.
Usage methodology terms like titration, subcutaneous injection, and stacking all show up in protocol discussions constantly, but they often get glossed over in favor of chasing the next peptide name. That's a mistake.
Subcutaneous injection (abbreviated as SubQ) means injecting into the fatty tissue just below the skin, typically in the abdomen, thigh, or flank. This is the most common administration route for most peptides because it allows for slower, more consistent absorption compared to intramuscular injection. Some peptides like BPC-157 are also used via intramuscular injection when targeting a specific muscle group or injury site, and the BPC-157 workflow changes depending on which route you choose.
Here's a numbered breakdown of safe stacking and titration practice:
- Start with a single peptide. Before stacking, establish a baseline with each compound individually so you can attribute any response, positive or negative, to the right source.
- Set your starting dose conservatively. For GLP-1 agonists, this means beginning well below the target therapeutic dose. For growth hormone secretagogues, a lower microgram range gives your pituitary time to respond without desensitizing.
- Hold each dose for 2 to 4 weeks. This gives you enough data to evaluate tolerance and efficacy before escalating.
- Introduce the second peptide only after stabilizing on the first. Stacking two unfamiliar compounds simultaneously makes it nearly impossible to troubleshoot side effects.
- Log everything. Dose, timing, route, sleep quality, energy, and any adverse reactions. You need this data to adjust intelligently rather than guessing.
- Taper or cycle off strategically. Many peptides downregulate receptors with continuous use. Planned breaks are part of the protocol, not an afterthought.
"The Wolverine stack is a great example of synergistic recovery stacking, but users who jump straight to full therapeutic doses of both BPC-157 and TB-500 simultaneously often misattribute improvements or side effects. Start low, confirm tolerance, then combine. Advanced stacking is a skill, not just a recipe." — Protocol methodology note from clinical peptide practitioners
Peptide protocol examples built around these steps produce far more consistent outcomes than protocols copied directly from forums without personal calibration.
Pro Tip: When titrating GLP-1 peptides like semaglutide, monitor GI symptoms closely during the first 4 to 8 weeks. Nausea and slowed gastric motility are not signs to push through aggressively. Slow the titration, and you'll retain far more of the intended metabolic benefit without compromising quality of life or adherence. And for performance peptide strategies beyond weight management, this same measured approach applies across the board.
Regulatory and safety terms everyone should know
Knowing how to use peptides is vital, but understanding how legal status and safety terminology impact you may be even more important.
Three terms you need to internalize before buying or using any peptide: FDA-approved, research peptide, and compounded.
FDA-approved therapeutic peptides have passed rigorous Phase I, II, and III clinical trials for a specific indication. Semaglutide is approved as Ozempic for type 2 diabetes and as Wegovy for obesity. These carry known safety profiles, standardized manufacturing, and legal prescribing pathways. They are not the same as what you order from a research vendor.
Research peptides carry labels explicitly stating "not for human use" or "for laboratory research only." This language is legally protective for the seller, not medically reassuring for you. Research peptides are not subject to the same manufacturing standards as pharmaceutical products. Contamination, incorrect concentrations, and mislabeling are documented risks in this supply chain. The majority of safety incidents in the peptide space trace back to research compounds, not approved therapeutics. This isn't a minor distinction.
Compounded peptides occupy a middle ground under 503A (patient-specific compounding by licensed pharmacies) and 503B (larger-scale outsourcing facilities subject to FDA oversight). Compounding allows access to peptides like BPC-157 through a prescriber when a commercially available product doesn't exist or isn't suitable, but regulatory scrutiny here has increased sharply. Always verify the pharmacy's accreditation status.
Key buyer and user scenarios based on regulatory status:
- Using an FDA-approved GLP-1 via prescription: Legally sound, well-studied safety profile, insurance may cover it
- Sourcing compounded BPC-157 through a 503A pharmacy with a prescriber: A regulated gray zone with more oversight than open research vendors
- Purchasing research peptides from an online vendor: High variability in purity and concentration, limited legal protection, all risk absorbed by you
- Stacking unapproved compounds without clinical oversight: Potentially effective based on preclinical data, but adverse events are entirely self-managed
For context on what the research foundation actually looks like, peptide safety research and synthetic peptide safety both offer rigorous breakdowns of where evidence is strong and where gaps remain.
Our perspective: where peptide terminology trips up even advanced users
Now that you're equipped to spot and understand core industry terms, here's what even long-time users often overlook.
The most experienced peptide users we encounter tend to fall into a specific trap. They master the nomenclature. They can rattle off half-lives, explain titration, and discuss stacking rationale with confidence. But they conflate the vocabulary of efficacy with the evidence of efficacy. These are not the same thing.
Knowing that BPC-157 and TB-500 are often stacked for synergistic recovery is useful. Understanding that both compounds lack human clinical trial data and that the preclinical results, while promising for tendon and joint repair, haven't been replicated in controlled human studies, is essential context that changes your risk calculus entirely.
The second failure pattern is letting social media terminology override regulatory language. Someone on a forum calls a compound "therapeutic-grade" because it came from a specific vendor. That phrase means nothing legally. It's not a regulatory classification. Users who adopt forum vocabulary as factual vocabulary end up making sourcing and dosing decisions based on community consensus rather than verifiable standards.
The lesson here isn't pessimism about peptides. It's calibration. Use authoritative sources for definitions. Cross-check regulatory status through official databases, not vendor copy. Stay current because this landscape shifts fast, and what was compoundable last year may be newly restricted this year. And explore peptide efficacy challenges so you understand what the research can and can't tell you before you commit to a protocol.
Pro Tip: Before starting any new peptide regimen, verify its current regulatory and clinical status through FDA databases or a licensed prescriber. Terminology on vendor sites reflects marketing intent, not clinical reality.
Get expert guidance for smarter peptide use
For readers serious about translating clear terminology into real fitness or longevity gains, here's where you can ensure the next phase is evidence-based and user-friendly.
Understanding terms is the foundation. Applying them consistently and safely across a personalized protocol is where Peptide AI comes in.
Peptide AI gives you a catalog of 50+ peptides with evidence-backed dosing parameters, an AI Insights Chatbot that answers real-time protocol questions with research citations, and an AI Body Scanner that tracks physical changes over time so your results aren't anecdotal. The app integrates with Apple Health, Oura Ring, and Whoop so your biometric data informs every decision. Whether you're titrating a GLP-1 for the first time or building a recovery stack around BPC-157 and TB-500, Peptide AI helps you move from terminology clarity to protocol precision. Available on iOS and Android.
Frequently asked questions
What does lyophilized mean, and why does it matter for peptide users?
Lyophilized refers to a peptide in freeze-dried powder form, which maximizes stability until it's reconstituted for use. If your peptide arrives in any other form without explanation, that's a quality concern worth investigating before injection.
How do I know if a peptide is FDA-approved or just for research?
FDA-approved peptides are classified as therapeutic, require a prescription, and are dispensed through licensed pharmacies. Research peptides explicitly state they are not for human use, which is a legal disclaimer, not a safety guarantee.
What is a "Wolverine stack" and who should use it?
The Wolverine stack combines BPC-157 and TB-500 targeting enhanced tissue healing through complementary pathways. Most supporting evidence is preclinical, and it's best approached by experienced users with clinical oversight rather than beginners.
Why do peptide protocols emphasize titration?
Titration gradually increases dose to minimize side effects and identify the lowest effective threshold before escalating, which is especially critical with GLP-1 agonists where GI side effects can be severe at higher starting doses.
