Synthetic peptides are widely dismissed as either a niche research tool or just another fitness supplement. That framing misses the bigger picture entirely. These lab-engineered molecules are actively reshaping regulated medicine, powering billion-dollar drug pipelines, and showing up in the wellness protocols of serious biohackers and clinicians alike. This guide cuts through the noise to explain what synthetic peptides actually are, how they're manufactured, what quality and safety really mean, and where the evidence genuinely supports their use. Whether you're new to peptide therapy or already running a stack, what follows will sharpen your thinking.
Table of Contents
- What are synthetic peptides?
- How are synthetic peptides made?
- Quality control and safety: What you need to know
- Therapeutic and wellness uses: Evidence, hype, and application
- The truth most guides miss: Quality and evidence matter more than hype
- How Peptide AI helps you navigate synthetic peptide therapy
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Synthetic peptide basics | Synthetic peptides are artificial chains of amino acids used in medicine, research, and wellness protocols. |
| Quality and safety first | Always check purity, manufacturing grade, and regulatory status before use to avoid health risks. |
| Evidence varies by use | FDA-approved peptides have strong support, while many wellness peptides lack robust human studies. |
| Production method matters | Production techniques impact the effectiveness, purity, and safety of peptides in real-world applications. |
What are synthetic peptides?
A peptide is simply a chain of amino acids. Your body makes thousands of them naturally, using them as hormones, signaling molecules, and structural components. Synthetic peptides are a different category: artificially produced amino acid chains of 2 to 50 residues, manufactured specifically for therapeutic, research, or wellness applications.
The key distinction from natural peptides isn't just origin. It's control. When a peptide is synthesized in a lab, chemists can precisely define its sequence, length, and modifications. That precision is impossible to replicate with peptides extracted from animal or plant sources, which carry variability, contamination risk, and batch inconsistency.
Synthetic peptides also differ from full proteins. Proteins are much longer chains, typically over 50 amino acids, and they fold into complex three-dimensional structures. Peptides are smaller, more targeted, and easier to engineer for a specific biological function. Think of a protein as a full toolkit and a peptide as the single precision instrument you actually need for one job.
The scope of this field is larger than most people realize. Over 100 FDA-approved peptide drugs exist today, and the global peptide therapeutics market exceeds $50 billion. That's not supplement territory. That's serious pharmaceutical infrastructure. For more on peptide basics and how they relate to wellness protocols, the landscape is worth understanding before you start any stack.
| Category | Origin | Regulatory status | Common uses |
|---|---|---|---|
| Natural peptides | Living organisms | Varies | Hormones, signaling |
| Synthetic peptides | Chemical synthesis | FDA-approved or research | Drugs, therapy, wellness |
| Research peptides | Lab synthesis | Unapproved | Investigation only |
| Compounded peptides | Pharmacy synthesis | Not FDA-approved | Off-label clinical use |
Main applications for synthetic peptides include:
- Pharmaceutical drugs: insulin, semaglutide, oxytocin
- Wellness and biohacking: BPC-157, TB-500, Semax
- Research tools: studying receptor binding, immune response
- Diagnostics: peptide-based assays and imaging agents
Understanding peptide synthesis overview at even a basic level helps you ask better questions about what you're actually putting into your body.
How are synthetic peptides made?
Production method matters more than most buyers realize. The same amino acid sequence can be produced with wildly different purity levels depending on the synthesis approach, equipment, and quality controls in place.

Solid-Phase Peptide Synthesis (SPPS) is the dominant method, capable of producing peptides up to 100 amino acids in length. It works by anchoring the growing peptide chain to a solid resin support, adding one protected amino acid at a time, then cleaving and purifying the finished product.
Here's how the SPPS process works step by step:
- Attach the first amino acid to a solid resin support
- Deprotect the terminal group to allow the next amino acid to bond
- Couple the next protected amino acid using activating reagents
- Repeat the deprotect-couple cycle for each residue in the sequence
- Cleave the completed peptide from the resin using acid
- Purify using High-Performance Liquid Chromatography (HPLC)
- Lyophilize (freeze-dry) the final product for stability and storage
Two alternative methods for peptide creation exist alongside SPPS:
| Method | Best for | Scale | Typical use |
|---|---|---|---|
| SPPS | Short to medium peptides | Small to medium | Research, pharma, wellness |
| Solution-phase synthesis | Simple, short sequences | Large industrial | Commodity peptides |
| Fragment condensation | Long or complex peptides | Medium | Complex therapeutics |
For peptide synthesis techniques used in wellness and therapeutic contexts, SPPS dominates because of its flexibility and precision.
Pro Tip: Ask your supplier whether modifications like PEGylation (attaching polyethylene glycol chains) or lipidation (adding fatty acid groups) were applied. These chemical modifications extend a peptide's half-life in the body, which directly affects dosing frequency and therapeutic effectiveness. A supplier who can't answer this question clearly is a red flag.
Quality control and safety: What you need to know
Here's where most wellness conversations about peptides fall short. Purity isn't a marketing term. It's a measurable, testable, critical safety variable.
Common impurities in synthetic peptides include:
- Deletion sequences: amino acids skipped during synthesis
- Truncated peptides: chains that terminated early
- Racemization: amino acids flipped to the wrong mirror-image form
- Oxidation: chemical degradation of sensitive residues
- Aspartimide formation: a side reaction that alters sequence integrity
These aren't theoretical concerns. Impurities can trigger immune responses, reduce efficacy, or introduce unpredictable biological activity. Research-grade peptides require over 98% purity, and GMP (Good Manufacturing Practice) grade is even more stringent, requiring sterility testing, endotoxin limits, and full batch documentation.
Research-grade peptides must meet a purity threshold of >98%. GMP-grade products go further, requiring sterility, endotoxin testing, and documented manufacturing controls. The difference between these grades isn't cosmetic. It's the difference between a controlled substance and an unknown compound.
Regulatory testing relies on two primary tools: HPLC (High-Performance Liquid Chromatography) confirms purity percentage, while Mass Spectrometry (MS) verifies the peptide's molecular identity. Without both, you can't confirm what you have.
The compounding vs. FDA-approved distinction matters enormously here. Compounded peptides aren't subject to the same pre-market review, meaning quality and consistency can vary significantly between batches and suppliers.
Warning signs of poor-quality peptides:
- No certificate of analysis (CoA) available
- Purity listed below 98% or not listed at all
- No HPLC or MS data provided
- Supplier can't confirm GMP or research-grade manufacturing
- Pricing significantly below market rate
- No sterility or endotoxin testing data
For more on synthesis challenges and risks at the manufacturing level, the research literature is detailed and worth reading before you commit to any source.
Pro Tip: Always request a CoA before purchasing. If a supplier hesitates or provides a generic document without batch-specific data, that's your answer.
Therapeutic and wellness uses: Evidence, hype, and application
Not all peptide claims are created equal. The gap between what's proven in clinical trials and what gets promoted in wellness communities is significant, and knowing the difference protects you.
On the firmly evidence-backed side: FDA-approved peptides like insulin and semaglutide have extensive human trial data supporting their safety and efficacy. Semaglutide alone has reshaped obesity and diabetes treatment globally.
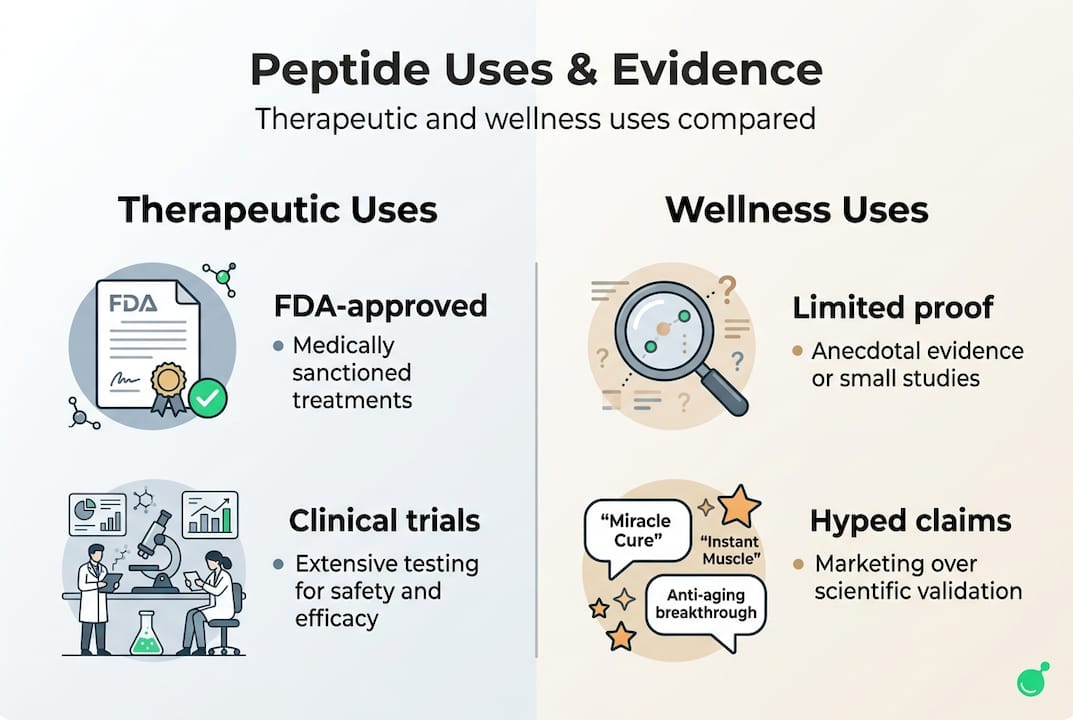
On the emerging side: TCP-25 showed a 76% reduction in wound inflammation in early trials, and TEIPP24 has shown promise in immune enhancement research. These are real signals, but they're early-stage.
Common peptide types and their primary applications:
- Insulin and GLP-1 agonists (semaglutide): metabolic disease, obesity
- BPC-157: gut repair, tissue healing (preclinical data only)
- TB-500: recovery and injury repair (animal studies)
- Semax: cognitive enhancement (limited human data)
- Growth hormone secretagogues (ipamorelin, CJC-1295): body composition
- Antimicrobial peptides: infection treatment (emerging research)
For AI-guided peptide protocols that help you track and optimize your stack based on real biometric data, the difference between evidence tiers matters for how you set expectations.
Potential risks and side effects to take seriously:
- Immunogenicity: your immune system may react to a foreign peptide sequence
- Cancer promotion: some peptides that stimulate growth pathways could theoretically accelerate tumor growth
- Contaminants: impure products introduce unpredictable biological effects
- Hormonal disruption: growth-related peptides can affect endogenous hormone balance
- Injection site reactions: common with subcutaneous administration
The modifications used in peptide therapy like PEGylation and lipidation help address some of these concerns by improving stability and reducing immunogenicity. Still, peptides for muscle and longevity remain an area where enthusiasm frequently outpaces the clinical evidence base.
The truth most guides miss: Quality and evidence matter more than hype
Most peptide guides focus on what a compound might do. Far fewer ask whether the product you're actually holding is what the label claims. That's the real conversation we need to have.
Wellness culture has a tendency to treat preclinical animal data as proof of human benefit. It isn't. Compounding risks and evidence gaps require genuine vigilance, not just a disclaimer buried in a blog post. The peptides showing the most social media traction, BPC-157 and TB-500 among them, still lack robust randomized controlled trial data in humans.
The overlooked variable is always quality. A peptide with 94% purity isn't a slightly inferior version of a 99% pure product. The 6% is an unknown mixture of deletion sequences, oxidized fragments, and synthesis byproducts, each with its own unpredictable biological activity.
If you're serious about peptide therapy, prioritize FDA-approved options first, use expert-guided protocols for anything else, and never buy from a source that won't show you batch-specific quality documentation. A personalized peptide protocol built on verified data is the only kind worth running.
Pro Tip: If a supplier can't provide documented purity of 98% or higher with batch-specific HPLC data, walk away. The savings aren't worth the risk.
How Peptide AI helps you navigate synthetic peptide therapy
Understanding synthetic peptides is step one. Applying that knowledge safely and effectively is where most people need support.
Peptide AI brings AI-powered protocol management, biometric tracking, and peer-reviewed research together in one mobile platform. Whether you're tracking BPC-157 for recovery, running a growth hormone secretagogue stack, or just starting to explore what peptide therapy can do for your biology, the app gives you real-time, data-backed guidance. With wearable integration, an AI Insights Chatbot, and a catalog of 50+ peptides, Peptide AI puts research-grade intelligence in your pocket. Available on iOS and Android.
Frequently asked questions
Are synthetic peptides safe for long-term use?
FDA-approved peptides are generally safe with well-documented safety profiles, but research-grade or compounded products may carry unknown long-term risks, so specialist oversight is essential.
How do I verify the quality of a synthetic peptide?
Request a batch-specific certificate of analysis showing purity and manufacturing grade with HPLC and MS data; GMP-grade products offer the highest assurance.
Can synthetic peptides really enhance muscle growth or healing?
Preclinical trials show potential for muscle repair and wound healing, but large-scale human studies are limited outside of FDA-approved drugs, so manage expectations accordingly.
What is the difference between synthetic, natural, and research peptides?
Synthetic vs. natural peptides differ primarily in origin and control: synthetic are lab-made with defined sequences, natural come from living organisms, and research peptides are unapproved compounds used for scientific investigation only.
Why do peptide protocols need medical or expert guidance?
Improper dosing, low-quality sources, and unknown interactions can cause real harm; expert oversight is strongly urged to ensure safe, monitored use with appropriate quality controls in place.
