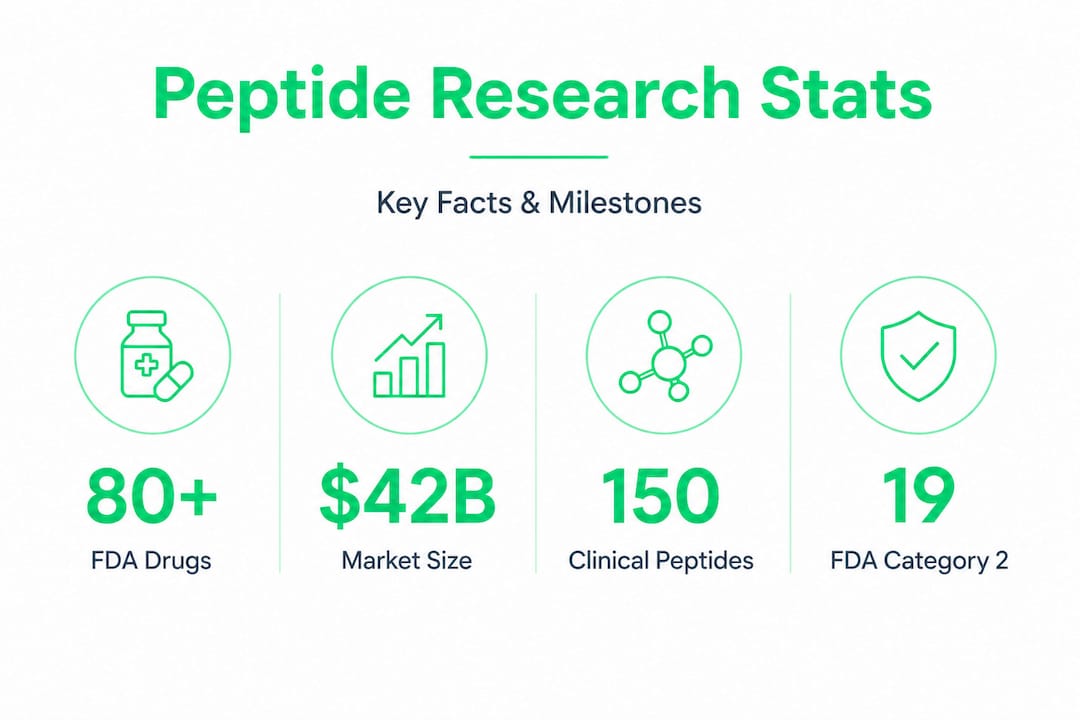
The peptide field has exploded from niche medical science into mainstream conversation, with over 80 approved drugs and a market projected to hit $80.4 billion by 2032. Athletes inject BPC-157 between training sessions, biohackers stack Semax for cognitive performance, and clinics offer GLP-1 analogs to millions. But the enthusiasm runs far ahead of public understanding. Most people engaging with peptide therapy today have a remarkably incomplete picture of what the research actually says, how these molecules are made, and where the serious safety lines are drawn. This article closes that gap.
Table of Contents
- Understanding peptide research: Definition and core goals
- How peptide research is performed: Synthesis and key techniques
- From lab to therapy: Peptide drugs, categories, and real-world application
- Safety, risks, and regulation: What every user should know
- The uncomfortable truth about peptide research most don't tell you
- Next steps with peptide research: Personalized insights and safe approaches
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Not all peptides are equal | FDA-approved peptides have strict safety evidence, while many research-only options lack solid clinical data. |
| Rigorous methods matter | Advanced synthesis and validation techniques underpin the quality and safety of therapeutic peptides. |
| The industry is booming | Global interest and investment in peptide therapies are driving rapid innovation and expanding treatment options. |
| Stay evidence-driven | Prioritize approaches validated in human studies and be wary of unapproved compounds. |
Understanding peptide research: Definition and core goals
Peptide research is the systematic scientific study of short amino acid chains (typically 2 to 50 amino acids) with the goal of identifying therapeutic, performance, and diagnostic applications. It spans everything from basic biochemistry in university labs to Phase III clinical trials in global pharmaceutical programs. The work covers synthesis, stability testing, bioavailability optimization, mechanism of action mapping, and safety profiling.
Why does this matter to you as someone exploring peptide therapy? Because the word "peptide" now covers an enormous spectrum. Insulin, a well-characterized peptide hormone, has decades of rigorous human evidence. BPC-157, popular in recovery circles, has compelling animal data but limited controlled human trials. These are not the same category of evidence, and treating them as equivalent is where things get dangerous.
The goals of peptide research include:
- Drug development: Creating new treatments for metabolic disease, cancer, cardiovascular conditions, and neurological disorders
- Recovery and regeneration: Identifying compounds that accelerate tissue repair and reduce inflammation
- Longevity research: Studying peptides that influence aging pathways, including growth hormone secretagogues
- Performance optimization: Investigating how peptides affect muscle protein synthesis, fat metabolism, and endurance
The fitness and biohacking communities have embraced peptides with genuine enthusiasm, but as peptide therapy evidence makes clear, the depth of human clinical data varies enormously between compounds. The rise of peptide therapeutics in metabolic, regenerative, and longevity medicine is real. The challenge is that fitness and biohacking use consistently outpaces what peer-reviewed human evidence can yet support.
"The most important question isn't whether a peptide works in rodents. It's whether it works safely in humans at therapeutic doses, and for most research-use-only compounds, that answer is still unknown."
Pro Tip: When evaluating any peptide, ask one simple question first: is there Phase II or Phase III human trial data? If the answer is no, you are working with preliminary science, not established therapy.
How peptide research is performed: Synthesis and key techniques
Understanding how peptides are made and validated helps you evaluate the quality of what you're using. The process is not a simple extraction from natural sources. It's a precise, multi-step technical workflow.
-
Solid-phase peptide synthesis (SPPS): Researchers build peptide chains amino acid by amino acid while anchored to a solid resin. The most common strategy is the Fmoc/tBu approach, which allows precise control of the sequence. Each step involves coupling reactions, washing, and deprotection. A single error at any stage can corrupt the final molecule.
-
Cleavage and deprotection: Once the chain is fully assembled, it is cleaved from the resin and protective chemical groups are removed. The resulting crude peptide contains the target molecule along with synthesis byproducts and incomplete sequences.
-
Purification via reverse-phase HPLC: High-performance liquid chromatography (HPLC) separates the target peptide from impurities based on polarity and molecular weight. Quality research laboratories achieve ≥95% purity using this method. Anything below this benchmark raises serious concerns about what else is in the vial.
-
Characterization via mass spectrometry: Mass spectrometry confirms the exact molecular weight and structure of the synthesized peptide. It catches truncated sequences, oxidized residues, and mismatched amino acids that HPLC alone might miss.
-
Analytical HPLC and amino acid analysis: A second analytical HPLC run verifies purity post-synthesis, and amino acid analysis confirms the exact composition of the peptide chain. These steps are standard in legitimate pharmaceutical-grade research.
| Stage | Method | Purpose | Quality benchmark |
|---|---|---|---|
| Synthesis | SPPS (Fmoc/tBu) | Build amino acid sequence | Correct chain assembly |
| Purification | Reverse-phase HPLC | Remove impurities | ≥95% purity |
| Identification | Mass spectrometry | Confirm molecular identity | Exact mass match |
| Verification | Analytical HPLC | Final purity check | Consistent peak profile |
| Composition | Amino acid analysis | Confirm sequence accuracy | Match to expected ratio |
Understanding peptide classifications decoded helps connect this technical workflow to practical therapy choices. When a supplier cannot provide a certificate of analysis showing mass spec data and HPLC purity, walk away.
Pro Tip: Always request a certificate of analysis (CoA) from any peptide supplier. It should show both HPLC purity and mass spectrometry confirmation. No CoA means no verified quality, regardless of what the label says.
From lab to therapy: Peptide drugs, categories, and real-world application
The translation from laboratory synthesis to real-world therapy is a long, regulated process. Yet the results have been extraordinary. Peptide-based drugs now treat type 2 diabetes (GLP-1 agonists like semaglutide), cancer (peptide receptor radionuclide therapy), cardiovascular disease (natriuretic peptides), and rare metabolic conditions. The pipeline is packed.

The global peptide drug market was valued at $42 billion in 2023, with approximately 150 peptides currently in clinical trials and over 400 in preclinical development. That scale reflects decades of foundational research now reaching maturity. The drugs you hear about most in medical news, including Ozempic, Wegovy, and their GLP-1 class, are direct products of this research pipeline.
| Category | Examples | Regulatory status | Evidence level |
|---|---|---|---|
| Approved drugs | Semaglutide, insulin, teriparatide | FDA approved | Phase III + post-market |
| Clinical trials | Novel GLP-1 variants, peptide vaccines | Active investigation | Phase I to III |
| Preclinical | Many recovery and CNS peptides | Research use only (RUO) | Animal models |
| Biohacker use | BPC-157, TB-500, Semax | Not FDA approved | Limited human data |
The therapeutic areas attracting the most peptide research investment right now include:
- Metabolic health: GLP-1, GIP, and dual agonists for obesity and insulin resistance
- Musculoskeletal recovery: Growth hormone secretagogues (ipamorelin, CJC-1295) and tissue repair compounds
- Cognitive performance: Nootropic peptides like Semax and Selank, studied primarily in Eastern European research
- Longevity and aging: Epithalamin, thymalin, and other bioregulator peptides with emerging mechanisms
Understanding synthetic peptide benefits puts this pipeline in context. The approved drugs represent proven value. The preclinical compounds represent potential value. The gap between those two is where most fitness and biohacking use currently sits.
Safety, risks, and regulation: What every user should know
Here is where the conversation gets uncomfortable for some people. The same properties that make peptides exciting, their biological specificity and potency, also create real risks when those molecules are used without adequate human safety data.
In 2023, the FDA placed 19 peptides in Category 2 of its compounding restrictions, citing immunogenicity risks (the tendency to trigger immune responses), limited human data, and documented adverse events including deaths in clinical studies. This wasn't regulatory overcaution. It was a response to real harm in real people.
Immunogenicity is the most underappreciated risk in peptide use. When your immune system recognizes a synthetic peptide as foreign, it can generate antibodies against it. In some cases, those antibodies cross-react with endogenous proteins, meaning your body starts attacking its own natural peptides. The severity can range from mild inflammation to serious autoimmune-like responses.
Other documented risks include:
- Injection site reactions: Inflammation, nodule formation, and tissue damage from improper preparation or contaminated product
- Hormonal disruption: Growth hormone secretagogues can alter the natural pulsatile release of GH, with unknown long-term consequences for the pituitary axis
- Cardiovascular effects: Some peptides affect heart rate and blood pressure, which matters especially for those already on cardiac medications
- Contamination risks: Research-grade peptides sourced outside pharmaceutical supply chains may contain endotoxins, solvents, or incorrect concentrations
- Lack of long-term data: Most biohacker-popular peptides have not been studied beyond weeks or months in humans
Before considering any peptide, peptide efficacy evidence should be your first stop. And for those just starting, best peptides for beginners focuses specifically on compounds with the most favorable evidence-to-risk profiles.
Safety checkpoints before considering peptide use:
- Confirm FDA or equivalent regulatory approval, or at minimum, active Phase II/III trial status
- Verify the compound has published human safety data, not just animal studies
- Obtain a certificate of analysis from any supplier before purchase
- Consult a physician familiar with peptide pharmacology, not just a general practitioner
- Understand the compound's mechanism and potential interactions with your current medications or conditions
"Regulatory approval is not a barrier invented by bureaucrats. It represents hundreds of thousands of data points confirming a compound's benefit-to-risk ratio in actual human beings."
The uncomfortable truth about peptide research most don't tell you
Here is what most peptide content online won't say directly: the fitness and biohacking community is running a large, uncontrolled human experiment, and the long-term results aren't in yet.
That's not a moral judgment. Many people in this space are thoughtful, data-driven individuals who have done more self-research than their doctors. The problem is that self-research, no matter how thorough, cannot substitute for properly controlled trials with statistical power, diverse populations, and years of follow-up. Individual anecdotes, even thousands of them, carry a different weight than randomized controlled data.
The peptide research landscape is genuinely exciting, especially for metabolic health and regenerative medicine. GLP-1 agonists are among the most significant medical developments of the last decade. The science is real. The promise is real. The problem is that the enthusiasm for approved, proven peptides bleeds over into enthusiasm for research-only compounds that share the name "peptide" but not the evidence base.
Consider the pattern: a peptide shows remarkable effects in rodent models. Online communities pick it up, suppliers start producing it as a research chemical, and within months people are self-injecting based on rat data. Years later, when human trials finally happen, the results are often more modest, more complicated, or sometimes reveal risks that animal studies missed entirely. This is not hypothetical. It is the documented history of many compounds that cycled through the biohacking world.
The practical takeaway here is not to avoid peptides. It's to apply a simple tiering system to your decision-making. Approved drugs with robust human data get the most trust. Active clinical trials with published interim safety data occupy the next tier. Preclinical compounds with only animal evidence belong in a separate, much higher risk category. Knowing which tier your compound sits in should shape everything from your decision to try it to the dose you consider and the monitoring you build around it.
For anyone serious about performance peptide examples, the most valuable skill you can develop isn't finding the next cutting-edge compound. It's accurately evaluating the evidence tier of whatever you're already using.
Next steps with peptide research: Personalized insights and safe approaches
Navigating the peptide landscape on your own is genuinely difficult. The research is scattered across dozens of journals, regulatory updates happen frequently, and the quality of online information varies from excellent to dangerously misleading. Getting this right requires personalized, data-backed guidance that keeps pace with evolving science.
Peptide AI was built specifically for this challenge. The app gives you access to a catalog of 50+ peptides, each backed by peer-reviewed research, with an AI Insights Chatbot that provides real-time, evidence-based recommendations tailored to your goals and biometrics. Whether you're tracking recovery with BPC-157, monitoring body composition changes with an AI Body Scanner, or syncing your wearable data from Oura Ring or Whoop, Peptide AI puts research-grade intelligence in your hands. It's not a substitute for medical supervision, but it is the most powerful tool available for staying informed, organized, and protocol-driven as you explore this field.
Frequently asked questions
What is the main purpose of peptide research?
Peptide research aims to develop safer, more effective therapies for metabolic, regenerative, and neurological conditions, with over 80 approved drugs already in clinical use and hundreds more in development.
Are all peptides safe for human use?
No. The FDA placed 19 peptides in Category 2 in 2023 due to immunogenicity risks and documented adverse events, including deaths, underscoring that many peptides lack adequate human safety data.
How do researchers ensure peptides are pure and safe?
Researchers use solid-phase peptide synthesis combined with reverse-phase HPLC purification and mass spectrometry confirmation to verify identity and achieve purity levels of at least 95%.
Why is there so much buzz about peptides among athletes and biohackers?
Peptides offer targeted mechanisms for performance, recovery, and longevity, but as fitness use outpaces evidence, many popular compounds still lack the controlled human trials needed to confirm both their benefits and long-term safety profile.
